THV Therapies Approved for Patients at High Risk for Aortic Valve Disease
More than 5 million Americans suffer from heart-valve disease. An estimated 1.5 million have aortic valve stenosis. Many patients with the dire diagnosis are fighting for their lives because of limited treatment options.
Until now, the sole treatment for aortic valve stenosis was open-heart surgery. A major operation, open-heart surgery involves putting the patient on a heart-lung machine while his or her valve is replaced. Patients who are too frail, have other ailments or are physically incapable of tolerating the procedure mostly likely will die two years after experiencing their first symptoms.
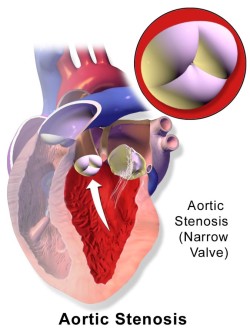
Aortic valve stenosis occurs when calcium deposits block openings and cause the heart to have to work harder in order to pump blood. It most often, but not always, is age-related and deadly if untreated. Onset is preceded by such conditions as arrhythmia, cardiac arrest, chest pain, fainting spells and heart failure.
Enter the transcatheter heart valve. The Food and Drug Administration has given the nod to a pair of THV systems that enable doctors to replace diseased heart valves arterially.
“These catheter-based therapies provide the only effective therapy for inoperable patients and are alternatives to open-heart surgery for patients at high risk for surgery,” wrote Dr. Jeffrey Shuren, of the FDA’s Center for Devices and Radiological Health, on the FDAVoice blog. “Both systems allow physicians to perform aortic valve replacement using catheters, so patients benefit from reduced symptoms of heart failure and improve their chances for survival.”
Each of the therapies is implanted via the leg, although other arteries can be used to deliver the replacement valve, depending on the patient’s condition. No heart-lung machine is necessary, making the associated risks exponentially lower.
The first device that gained expanded approved is CoreValve by Medtronic. The agency previously had OK’d the implant for inoperable patients only but now has done so for high-risk patients. The decision came after a 795-participant clinical trial showed a decreased mortality rate among those at high-risk who received the CoreValve product.
The second device made available is Sapien XT by Edwards Lifesciences. It is a balloon-expandable THV – an upgrade from the previous FDA-approved Sapien – that targets patients with diseased or problem arteries.
“There is a clear and important public health need for both these devices,” Shuren wrote. “With the additional availability of two THV systems, physicians will now be able to treat the widest possible spectrum of patients with severe and symptomatic aortic stenosis. They will be able to choose the procedure or device that best suits each individual patient based on unique patient characteristics, anatomic constraints, implant procedure risks, device characteristics, and overall benefit-risk profiles. After all, patients are at the core of what we do.”
Share This