Why is Potentially Lethal Contraceptive NuvaRing on the Market?
It’s always a good thing when a bad product gets called out by mainstream media. Unless you have been fitted with a NuvaRing birth control device, made by Merck, or know one of the thousands of litigants who have suffered serious or fatal blood clots, you may be blissfully ignorant of its dangers.
Vanity Fair’s writer-at-large Marie Brenner peels back the veil of ignorance on this dangerous birth-control device in its December 9, 2013 edition with the headline “Why is Potentially Lethal Contraceptive NuvaRing Still on the Market?” That’s a good question that many who handle mass tort claims have been asking for years.
The answer may be in the first paragraph. NuvaRing brought drugmaker Merck (maker of Vioxx) $623 million in profit in 2012.
Brenner profiles 24-year-old Erika Langhart who was found writhing on the floor gasping for air just before Thanksgiving in 2011. Hospitalized, she died a few days later of a pulmonary embolism. What was the one question the emergency room doctor had for her mother? “Was your daughter using birth control?” Erika had a NuvaRing implanted four years earlier. Her cause of death was listed as “Massive, Double Pulmonary Embolism – a direct result of the NuvaRing.”
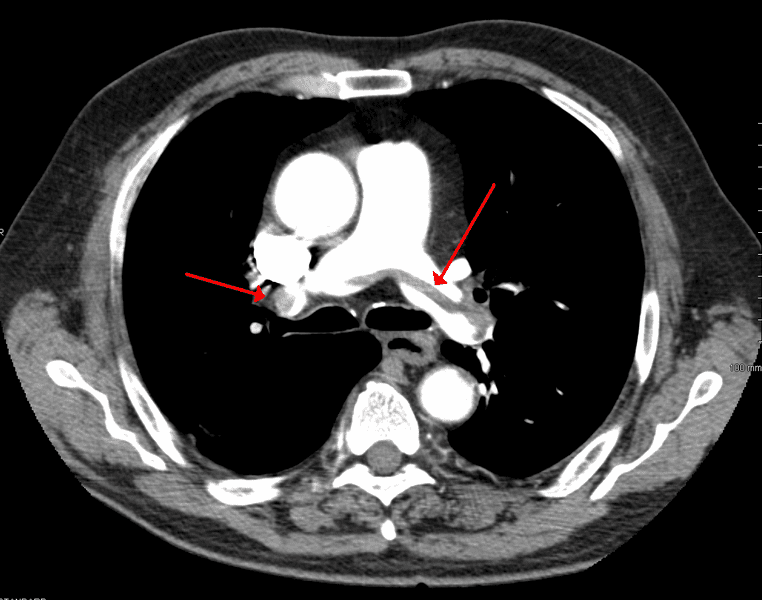
Another young woman collapsed 10 days after she started using NuvaRing. A CT scan showed she had developed dozens of blood clots in her lungs. Doctors credited her activities in an army elite team of athletes as the only reason she survived. She was told her career as an athlete was over.
The Food and Drug Administration (FDA) approved NuvaRing in 2002 as an alternative to birth control pills. The fitted ring is not unlike alternate hormone-releasing birth control methods which also include implants and skin patches. NuvaRing contains ethinyl estradiol (a synthetic version of estrogen) and etonogestrel (a synthetic progesterone) that’s released for three weeks. Compared to oral contraceptives containing levonorgestrel, the British Medical Journal (BMJ) reported the risk for a venous thromboembolism (VTE) was 1.9 times higher with the ring. An FDA study came to a similar conclusion that the risk for VTE was 1.56 times higher for NuvaRing users.
The Vanity Fair story reports that NuvaRing received approval despite paperwork submitted to the agency as backup showing the potential risks of developing blood clots. Omission from the 30-page approval summary virtually assures the FDA will not have the manpower to sort through hundreds of pages of backup documentation.
Canada may not be as lax in its regulatory environment. In Canada, the NuvaRing label includes a warning based on a study of more than a million women that showed NuvaRing recipients were six times more likely to develop blood clots when compared to other contraceptives.
Adverse Events, a company that tracks the FDA database for drug injuries, reports there have been 161 patient deaths associated with NuvaRing and 1,834 hospitalizations.
The company says compared to other contraceptives, NuvaRing has the fourth highest RXScore, an evaluation by Adverse Events that indicates its risk level. Only Prempro, Depo-Provera and Yasmin have slightly higher scores. RxScore is a proprietary algorithmic scoring based on safety data from over five million complication reports to the FDA.
Clearly from the response to this Vanity Fair article, the public does not know what is buried inside the FDA’s website. Thank you, many readers say in the comments including the mother of a young woman who died suddenly seven years ago of double pulmonary embolism despite the fact she had just had a complete physical and was with Military Sealift Command. Another woman asks how Vanity Fair can keep the actual article behind a pay wall when it is “too important to too large an audience.”
That sounds like a very good question.
Share This