FDA Revises Safety Recommendations for Popular Antidepressant Celexa
The U.S. Food and Drug Administration (FDA) has revised its recommendations for the popular antidepressant, Celexa (citalopram hydrobromide).
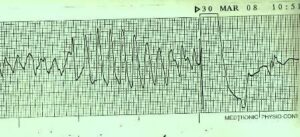
Last August, the FDA warned Celexa should be limited to doses no greater than 40 mg per day because of a risk of abnormal electrical activity in the heart known as Torsade de Pointes, which can be fatal.
The drug label was revised again Tuesday, March 27, to strengthen the warnings once again. The recommendations include:
In patients who suffer preexisting heart conditions, specifically, those who experience congenital prolonged QT intervals or who have suffered a recent heart attack, hypokalemia, bradycardia or hypomagnesemia, Celexa is “not recommended,” however, the FDA recognizes there are some people who may not have viable alternatives and may be able to take a lower dose of citalopram or Celexa. In those patients, the medical provider should monitor electrolyte levels and conduct ECG monitoring.
Patients who have a liver impairment should not take Celexa over the 20 mg per day dose.
Celexa is not recommended for any patient who is taking other drugs that also prolong the QT interval, which is the ECG measurement of the time between the ‘Q’ and ‘T’ wave in the heart’s electrical cycle. A faster heart fate, the shorter the QT interval.
Anyone over the age of 60, or with liver impairment, should be limited to the maximum recommended dose of 20 mg per day. Celexa is not recommended in patients also taking Tagamet, which can increase the level of Celexa in the blood, increasing the risk of Torsade de Pointes.
A Popular SSRI
Celexa is taken by about 7.2 million patients who received a prescription in 2011. The drug belongs to the class of antidepressants called SSRIs (selective serotonin reuptake inhibitors). Any patient taking more than the 40 mg dose should check with his doctor. The FDA recommends if a patient taking Celexa experiences dizziness or a lightheaded feeling, he should check with his medical professional who may want to do an ECG and check the patient’s potassium and magnesium levels.
The recommendations cover both the Forest Laboratories Inc’s Celexa and its generic version.
As we’ve reported here before, any pregnant woman taking an antidepressant needs to understand that medication taken during pregnancy may contribute to life-threatening birth defects. Celexa, Prozac, Paxil, Lexapro, Zoloft, Effexor and Pristiq all fall into the category of SSRIs.
Share This