CPAP Recall: Sleep Apnea Patients May Have an Alternative
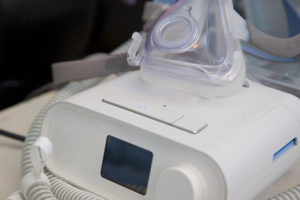
Nearly six months after Philips recalled millions of its CPAP, BiPAP, and ventilator machines, some patients have hope that they will receive a replacement machine. In one Massachusetts town, some sleep apnea patients who are in the higher risk category may have the option of receiving new CPAP machines free of charge.
Numerous insurance companies are waiving a rule that requires patients to wait at least five years before obtaining a new CPAP machine. Because CPAP machines are medically necessary and lifesaving for some patients, insurance companies are helping high-risk sleep apnea patients.
Obtaining a new machine is a much desirable option for patients who otherwise must use a Philips machine that is defective and potentially dangerous. For other patients, they may have no choice but to continue using Philips CPAP machines until the company replaces the foam on all machines with a safer alternative.
Why Were Philips CPAP Machines Recalled?
In June of 2021, the United States Food and Drug Administration (FDA) issued a recall of millions of Philips CPAP, BiPAP, and ventilator machines. The reason for the recall relates to the sound abatement foam used with the machines. The defective machines had a polyester-based polyurethane (PE-PUR) foam that degrades over time. Such degradation can cause serious injuries or illnesses to patients who inhale or ingest the degraded dust particles.
In a recent update on the CPAP recall, the FDA indicated that the replacement foam Philips has chosen, which is a silicone-based foam, has failed at least one safety test concerning the presence of volatile organic compounds (VOCs). Philips underwent similar testing, which did not yield the same negative results.
As such, the FDA has asked that Philips retain an independent laboratory to perform testing on the replacement silicone-based foam. Many patients have already suffered personal injuries from the defective PE-PUR foam that degrades, emitting potentially toxic substances into their airways. Waiting for a replacement product that is equally dangerous places patients in a precarious situation.
Injuries and Illnesses Associated with Philips CPAP Machines
The sound abatement foam used in recalled Philips CPAP, BiPAP, and ventilator machines can cause numerous serious injuries. Examples of such injuries include, but may not be limited to, the following:
- Acute Respiratory Distress Syndrome (ARDS)
- Breast cancer
- Heart attack
- Heart failure
- Brain cancer
- Kidney damage
- Kidney disease
- Bladder cancer
- Liver damage and/or disease
- Leukemia
- Upper respiratory inflammation
- Respiratory failure
Injuries will vary depending on a patient’s underlying health. However, the inhalation or ingestion of degraded particles of PE-PUR foam can be harmful to any person, regardless of existing underlying health conditions. Many patients who have suffered personal injuries are filing lawsuits in court, with tens of thousands of lawsuits (if not more) expected to come.
Contact Searcy Denney Today for a Free Consultation
If you were diagnosed with a medical condition, such as ARDS or cancer, after using a Philips CPAP, BiPAP, or ventilator machine subject to recall, it is time to consider speaking with a CPAP recall lawyer to discuss potential legal options. The defective medical device lawyers of Searcy Denney represent numerous CPAP clients who have been injured. To schedule a free consultation to speak with a CPAP recall lawyer, contact our office today by calling (800) 780-8607 or by completing a free case evaluation form on our website.
Share This


